- 首页 > 正文
ID Week前沿|真实世界研究:病毒抑制的经治HIV感染者B/F/TAF转换治疗较DTG/3TC的安全性更高
感染医线 发表时间:2023/10/23 20:07:30
编者按:比克替拉韦/恩曲他滨/丙酚替诺福韦(B/F/TAF)是目前治疗HIV感染最广泛应用的单片制剂方案,也是美国卫生与人类服务部(DHHS)推荐的适用于最广泛HIV感染人群的初始治疗方案,无需等待耐药检测结果即可启动治疗,且不受HBV合并感染、病毒载量和CD4+ T细胞计数的影响[1-3]。多替拉韦/拉米夫定(DTG/3TC)也是常见的单片制剂方案,但是对病毒载量、耐药检测和HBV血清学结果有要求,它仅适用于经耐药测试的、病毒载量(viral load,VL)≤500,000 copies/mL且不合并HBV感染的人群[2]。近年来开展的一系列随机对照研究(RCT)研究均证实,在已获得病毒学抑制的经治HIV感染者中,采用B/F/TAF[4-11]和DTG/3TC[12-15]方案进行转换治疗具有良好的抗病毒疗效和安全性。2023年10月10日至15日,全球规模最大的感染病学术会议之一——美国感染性疾病周(ID Week)在波士顿隆重举行。会上,美国佛罗里达州Vero Beach家庭健康中心Gerald Pierone Jr博士口头报告了一项真实世界研究(大会摘要号:1023)[16],比较了在已获得病毒学抑制的经治HIV感染者中,采用B/F/TAF和DTG/3TC方案进行转换治疗的病毒学失败(virologic failure,VF)风险和停药风险。其研究结果表明,与B/F/TAF相比,DTG/3TC的药物相关停药风险更高。
该项研究是一项基于美国大型队列 OPERA? 的分析,该队列记录了在美国 85家诊所接受治疗的超过155,000名HIV感染者的数据。研究纳入标准为:2020年8月1日至2022年6月30日期间,所有VL<200 copies/mL的、成年(≥18岁)经治HIV感染者,接受B/F/TAF或DTG/3TC方案进行转换治疗。研究将VF定义为:连续2次检测到VL≥200 copies/mL或VL≥200 copies/mL后停药;在深度的灵敏度分析中使用VL≥50 copies/mL为截断值;停药被定义为:任何对于转换方案的修改或治疗间隔>45天。采用倾向评分逆概率加权(IPTW)调整基线时的种族、CD4+ T细胞计数和eGFR等特征,估计VF发生率和危险比(HR)。
最终,研究纳入3512例接受B/F/TAF转换治疗的HIV感染者,中位随访16(四分位间距:11,22)个月;以及2327例接受DTG/3TC治疗的HIV感染者,中位随访15(四分位间距:10,21)个月。两组受试者的基线临床特征如表1所示,使用IPTW实现了组间平衡(见表1)。
表1. 两组受试者的基线临床特征
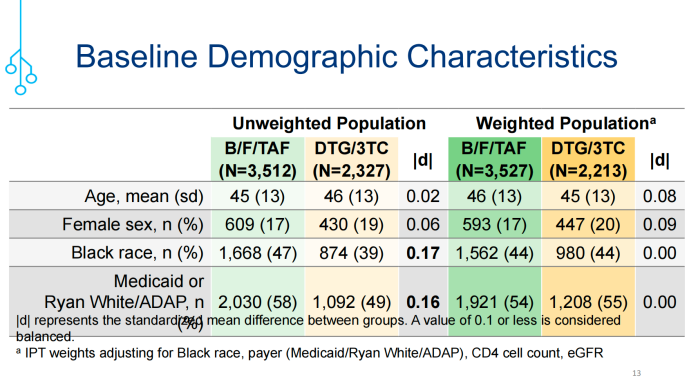
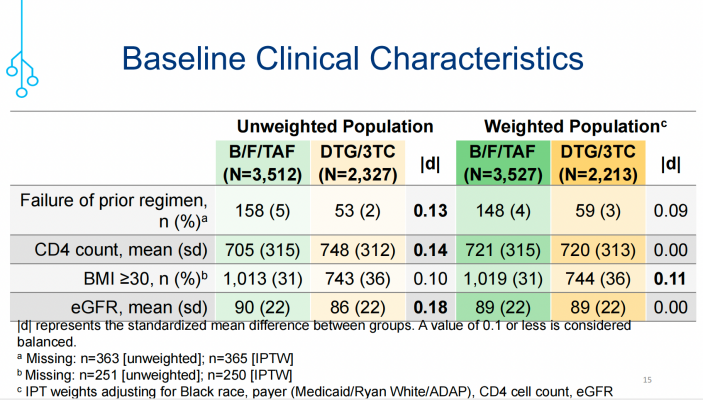
(引自讲者幻灯)
结果表明,在IPTW校正后采用VF≥200 copies/mL评估,B/F/TAF组和DTG/3TC组分别有2%(n=84)vs. 3%(n=59)出现VF,两组间的差异没有统计学意义(HR=0.84;95%CI:0.59-1.18);当采用VF≥50 copies/mL评估时,VF的发生率分别升高至9%(n=310)vs. 8%(n=179),但两组之间的风险差异仍没有统计学意义(HR=1.04;95%CI:0.86-1.26;图1)。
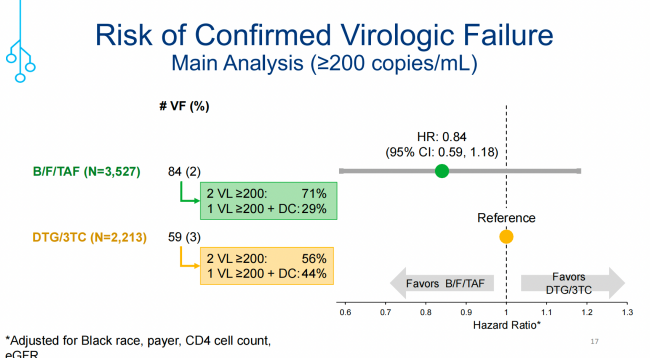
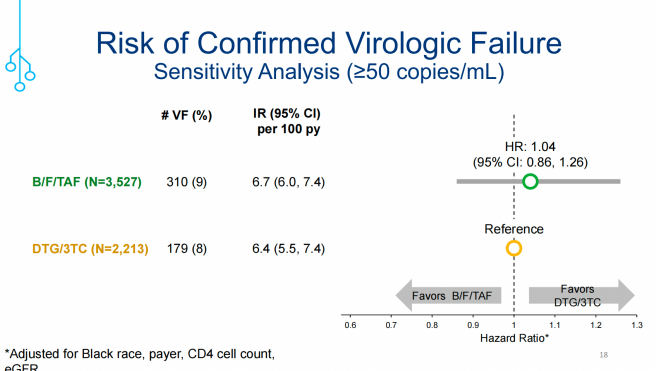
图1. 两组受试者的VF(≥200copies/mL和≥50 copies/mL)风险评估(引自讲者幻灯)
与DTG/3TC相比,B/F/TAF组停药的可能性较小(HR=0.83;95%CI:0.73-0.94;图2)。B/F/TAF组和DTG/3TC组中,分别有6%(n=38)vs. 9%(n=40)的发生与治疗相关的停药(图3),包括:最后一次检测VL≥200 copies/mL(3% vs. 5%)、不良反应(3% vs. 4%)、实验室检查指标异常(≤1% vs. ≤1%)。
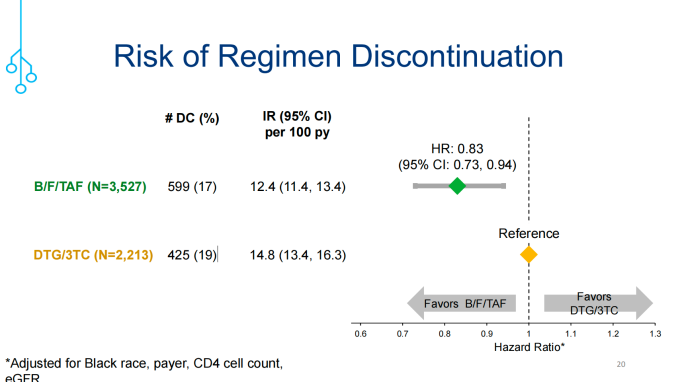
图2. 两组受试者的停药风险评估
(引自讲者幻灯)
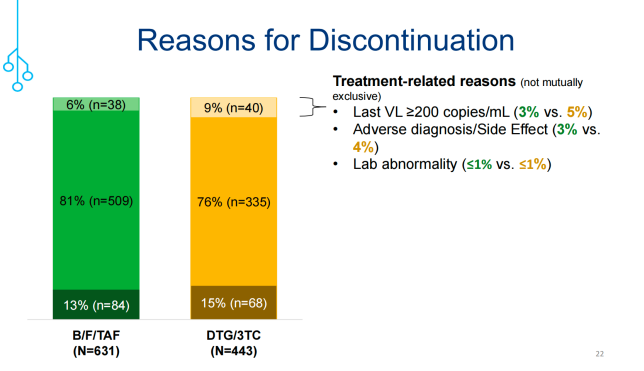
图3. 两组受试者发生药物相关停药比例及的原因分析(引自讲者幻灯)
总体来看,该项真实世界研究数据显示,在已获得病毒学抑制的经治HIV感染者中,采用B/F/TAF进行转换治疗者显著多于DTG/3TC转换者(61% vs. 39%)。在研究过程中,所有受试者中VF不常见,两种方案的VF风险相似;然而与B/F/TAF相比,DTG/3TC发生药物相关停药的例数更多。两组中大多数停药似乎与治疗的疗效或发生严重的安全性/耐受性事件无关。
参考文献
1. DHHS. Guidelines for the Use of Antiretroviral Agents in Adults and Adolescents Living with HIV. Accessed: March 2023. Available at: https://clinicalinfo.hiv.gov/sites/default/files/guidelines/documents/adult-adolescent-arv/guidelines-adult-adolescent-arv.pdf.
2. Gandhi R T, Bedimo R, Hoy J F, et al. Antiretroviral drugs for treatment and prevention of HIV infection in adults: 2022 recommendations of the International Antiviral Society–USA Panel[J]. JAMA, 2023, 329(1): 63-84.
3. EACS. Guidelines for treatment of people living with HIV version 11.1. Accessed: October 2022. Available at: https://www.eacsociety.org/media/guidelines-11.1_final_09-10.pdf.
4. Jain P, Morrisette T, Scherrer S, et al. Pharmacist-led patient satisfaction survey for people living with human immunodeficiency virus taking single-tablet bictegravir/emtricitabine/tenofovir alafenamide after being switched from twotablet dolutegravir plus emtricitabine/tenofovir alafenamide. In: Proceedings of the 2019 ACCP Annual Meeting, New York, 26-29 October 2019.
5. Tiraboschi J, Prieto P, Saumoy M, et al. Neuropsychiatric, clinical and laboratory changes in patients prospectively switching from elvitegravir/cobicistat/emtricitabine/tenofovir alafenamide to bictegravir/emtricitabine/tenofovir alafenamide. In: Proceedings of the HIV Glasgow 2020, Glasgow, 5-8 October 2020.
6. Rojas Lievano J, Inciarte A, De La Mora L, et al. Experience from a real-life cohort: Outcome of 984 HIV infected patients treated with bictegravir/emtricitabine/tenofovir alafenamide in Hospital Clinic, Barcelona. In: Proceedings of the HIV Glasgow 2020, Glasgow, 5-8 October 2020.
7. Doyle C, Moriarty M, Devitt E. Real-world experience of bictegravir (BIC)/emtricitabine (FTC)/tenofovir alafenamide (TAF) in a diverse urban HIV clinic cohort. HIV Med 2020; 21: 16–74.
8. Clark F, Underwood J, Lomax N. Clinical outcomes of stable patients switched to bictegravir/emtricitabine/tenofovir alafenamide - Biktarvy. HIV Med 2020; 21: 16–74.
9. Chamberlain N, Brock JB, Mena LA. Bic/FTC/TAF maintains viral suppression in patients with documented m184v/I mutations: A real world experience. Open Forum Infect Dis 2020; 7: S530.
10. Hayes E, Derrick C, Smalls D, et al. Short-term adverse events with BIC/FTC/TAF: postmarketing study. Open Forum Infect Dis 2020; 7: ofaa285.
11. Rolle CPM, Nguyen V, Cruz D, et al. Efficacy, safety and tolerability of switching to bictegravir/emtricitabine/tenofovir alafenamide (B/F/TAF) in HIV-1 infected virologically-suppressed older adults in a real-world setting. Open Forum Infect Dis 2020; 7: S534–S535.
12. Fabbiani M, Gagliardini R, Ciccarelli N, et al. Atazanavir/ritonavir with lamivudine as maintenance therapy in virologically sup- pressed HIV-infected patients: 96 week outcomes of a randomized trial. J Antimicrob Chemother. 2018;73(7):1955–1964.
13. Arribas JR, Girard PM, Landman R, et al. Dual treatment with lopinavir-ritonavir plus lamivudine versus triple treatment with lopinavir-ritonavir plus lamivudine or emtricitabine and a second nucleos(t)ide reverse transcriptase inhibitor for maintenance of HIV-1 viral suppression (OLE): a randomised, open-label, non-inferiority trial [published correction appears in Lancet Infect Dis. 2015 Aug;15(8):875]. Lancet Infect Dis. 2015;15(7):785–792.
14. Pulido F, Ribera E, Lagarde M, et al. Dual therapy with darunavir and ritonavir plus lamivudine vs triple therapy with darunavir and ritonavir plus tenofovir disoproxil fumarate and emtricitabine or abacavir and lamivudine for maintenance of human immunodeficiency virus Type 1 viral suppression: randomized, open-label, noninferiority DUAL-GESIDA 8014-RIS-EST45 trial. Clin Infect Dis. 2017;65 (12):2112–2118.
15. Perez-Molina JA, Pulido F, Di Giambenedetto S, et al. Individual patient data meta-analysis of randomized controlled trials of dual therapy with a boosted PI plus lamivudine for maintenance of virological suppression: GeSIDA study 9717. J Antimicrob Chemother. 2018;73(11):2927–2935.
16. Gerald Pierone, et al. Suppressed Switch to Bictegravir/Emtricitabine/Tenofovir Alafenamide vs. Dolutegravir/Lamivudine: Virologic Failure and Durability. IDWeek 2023 Abstract 1023.
往期推荐
来源:《感染医线》
声 明
凡署名原创的文章版权属《感染医线》所有,欢迎分享、转载(开白可后台留言)。本文仅供医疗卫生专业人士了解最新医药资讯参考使用,不代表本平台观点。该等信息不能以任何方式取代专业的医疗指导,也不应被视为诊疗建议,如果该信息被用于资讯以外的目的,本站及作者不承担相关责任。

责任编辑:彭伟彬
相关搜索: IDWeek 2023