- 首页 > 正文
一图读懂:一场隐秘而伟大的战争——人类与HIV的战斗历程丨世界艾滋病日
感染医线 发表时间:2023/12/5 19:17:39
根据联合国艾滋病规划署(UNAIDS)发布的《终结艾滋病之路:2023年全球艾滋病报告》,目前,全球现存有3900万HIV感染者,其中2980万正在接受抗反转录病毒治疗(ART)[1]。
自1981年,美国疾病预防控制中心(CDC)报道了第一批疑似因HIV感染导致机会性感染的病例后[2],HIV的流行史由此展开。时至今日,HIV感染已经由无法医治的疾病成为进程可控的慢性疾病,因此,HIV的流行史也是ART药物的发展史和人类与HIV的战斗史。
1988年12月1日,为提高对HIV感染和传播的认识,并悼念因艾滋病死亡患者的第一届世界艾滋病日活动启动,此后,每年的12月1日被设立为世界艾滋病日。
在2023年的世界艾滋病日即将到来之际,我们与大家共同回顾40年来,人类与HIV战斗中的里程碑事件,并分享未来ART药物发展和终结HIV流行的愿景。
▌参考文献:
1. Centers for Disease Control and Prevention. Pneumocystis pneumonia – Los Angeles. MMWR 1981;30:250–2. Available from:https://www.cdc.gov/mmwr/preview/mmwrhtml/lmrk077.htm (Accessed 1 May 2023).
2. Centers for Disease Control and Prevention. Current trends update on acquired immune deficiency syndrome (AIDS) – United States. MMWR 1982;31:507–8, 513–14. Available from: https://www.cdc.gov/mmwr/preview/mmwrhtml/00001163.htm (Accessed 1 May 2023).
3. Barre-Sinoussi F, Chermann J, Rey F et al. Isolation of a T-lymphotropic retrovirus from a patient at risk for acquired immune deficiency syndrome (AIDS). Science 1983;220:868–71.
4. Lee L, Karon J, Selik R et al. Survival after AIDS diagnosis in adolescents and adults during the treatment era, United States, 1984-1997. JAMA 2001;285:1308–15.
5. National Museum of American History. 1985. Abbott HTLV III EIA (Enzyme immunoassay for the detection of antibody to human T-lymphotropic virus type III in human serum or plasma). Available from:https://americanhistory.si.edu/collections/search/object/nmah_1322289 (Accessed 1 May 2023).
6. US Food and Drug Administration. The history of FDA's role in preventing the spread of HIV/AIDS. 2019. Available from: https://www.fda.gov/about-fda/fda-history-exhibits/history-fdas-role-preventing-spread-hivaids (Accessed 1 May 2023).
7. Fischl M, Richman D, Grieco M et al. The efficacy of azidothymidine (AZT) in the treatment of patients with AIDS and AIDS-related complex. A double-blind, placebo-controlled trial. N Engl J Med 1987;317:185–91.
8. Centers for Disease Control and Prevention. Current trends first 100,000 cases of acquired immunodeficiency syndrome – United States. MMWR 1989;38:561–3. Available from:https://www.cdc.gov/mmwr/preview/mmwrhtml/00001442.htm (Accessed 1 May 2023).
9. Centers for Disease Control and Prevention. Update: mortality attributable to HIV infection/AIDS among persons aged 25-44 years – United States, 1991 and 1992. MMWR 1993;42:869–72. Available from: https://www.cdc.gov/mmwr/preview/mmwrhtml/00022174.htm (Accessed 1 May 2023).
10. Concorde Coordinating Committee. Concorde: MRC/ANRS randomised double-blind controlled trial of immediate and deferred zidovudine in symptom-free HIV infection. Concorde Coordinating Committee. Lancet 1994;343:871–81.
11. Aboulker J, Swart A. Preliminary analysis of the Concorde trial. Concorde Coordinating Committee. Lancet 1993;341:889–90.
12. Connor E, Sperling R, Gelber R et al. Reduction of maternal-infant transmission of human immunodeficiency virus type 1 with zidovudine treatment. N Engl J Med 1994;331:1173–80.
13. Centers for Disease Control and Prevention. First 500,000 AIDS cases – United States, 1995. MMWR 1995;44:849–53. Available from:https://www.cdc.gov/mmwr/preview/mmwrhtml/00039622.htm (Accessed 1 May 2023).
14. Gulick R, Mellors J, Havlir D et al. Treatment with indinavir, zidovudine, and lamivudine in adults with human immunodeficiency virus infection and prior antiretroviral therapy. N Engl J Med 1997;337:734–9.
15. Wise J. HIV epidemic is far worse than thought. BMJ 1997;315:1486.
16. Palella F, Delaney K, Moorman A et al. Declining morbidity and mortality among patients with advanced human immunodeficiency virus infection. N Engl J Med 1998;338:853–60.
17. HIV.gov. A timeline of HIV and AIDS. 2022. Available from: https://www.hiv.gov/hiv-basics/overview/history/hiv-and-aids-timeline/#year-1999 (Accessed 1 May 2023).
18. Lucas G, Chaisson R, Moore R. Highly active antiretroviral therapy in a large urban clinic: risk factors for virologic failure and adverse drug reactions. Ann Intern Med 1999;131:81–7.
19. Henry K. The case for more cautious, patient-focused antiretroviral therapy. Ann Intern Med 2000;132:306–11.
20. UNAIDS. Global HIV & AIDS statistics – Fact sheet 2022. Available from: https://www.unaids.org/sites/default/files/media_asset/UNAIDS_FactSheet_en.pdf (Accessed 19 August 2022).
21. Sax P. Antiretroviral therapy: now "it just works". Clin Infect Dis 2011;53:605–8.
22. Hesselgrave BL. The 13th International AIDS Conference 2000: Durban, South Africa. J Public Health Manag Pract 2001;7:ix–xi.
23. Fleck F. WHO issues global alert after grim report on HIV/AIDS. BMJ 2003;327:698.
24. Gilead. ISENTRESS Product Information. May 2022. Available from: https://www.merck.com/product/usa/pi_circulars/i/isentress/isentress_pi.pdf (Accessed 1 May 2023).
25. Cohen M, Chen Y, McCauley M et al. Antiretroviral therapy for the prevention of HIV-1 transmission. N Engl J Med 2016;375:830–9.
26. Gilead Sciences. TRUVADA Product Information. June 2020. Available from: https://www.gilead.com/~/media/files/pdfs/medicines/hiv/truvada/truvada_pi.pdf (Accessed 23 December 2022).
27. UNAIDS. Global AIDS update 2016. Available from:https://www.unaids.org/sites/default/files/media_asset/global-AIDS-update-2016_en.pdf (Accessed 19 August 2022).
28. Nance R, Delaney J, Simoni J et al. HIV viral suppression trends over time among HIV-infected patients receiving care in the United States, 1997 to 2015: a cohort study. Ann Intern Med 2018;169:376–84.
29. INSIGHT START Study Group, Lundgren J, Babikar A et al. Initiation of antiretroviral therapy in early asymptomatic HIV infection. N Engl J Med 2015;373:795–807.
30. TEMPRANO ANRS 12136 Study Group, Danel C, Moh R et al. A trial of early antiretrovirals and isoniazid preventive therapy in Africa. N Engl J Med 2015;373:808–22.
31. Panel on Antiretroviral Guidelines for Adults and Adolescents. Guidelines for the use of antiretroviral agents in HIV-1-infected adults and adolescents, January 2016. Department of Health and Human Services. Available from: https://clinicalinfo.hiv.gov/sites/default/files/guidelines/archive/AdultandAdolescentGL003412.pdf (Accessed 9 December 2022).
32. Molina J, Ward D, Brar I et al. Switching to fixed-dose bictegravir, emtricitabine, and tenofovir alafenamide from dolutegravir plus abacavir and lamivudine in virologically suppressed adults with HIV-1: 48 week results of a randomised, double-blind, multicentre, active-controlled, phase 3, non-inferiority trial. Lancet HIV 2018;5:e357–65.
33. UNAIDS. Global AIDS Strategy 2021–2026 - End Inequalities. End AIDS. Available from: https://www.unaids.org/sites/default/files/media_asset/global-AIDS-strategy-2021-2026-summary_en.pdf (Accessed 9 December 2022).
34. Vitoria M, Rangaraj A, Ford N et al. Current and future priorities for the development of optimal HIV drugs. Curr Opin HIV AIDS. 2019;14:143–9.
35. The CASCADE Collaboration. Survival after introduction of HAART in people with known duration of HIV-1 infection. Lancet 2000;355:1158–9.
里程碑1. HIV流行的开端
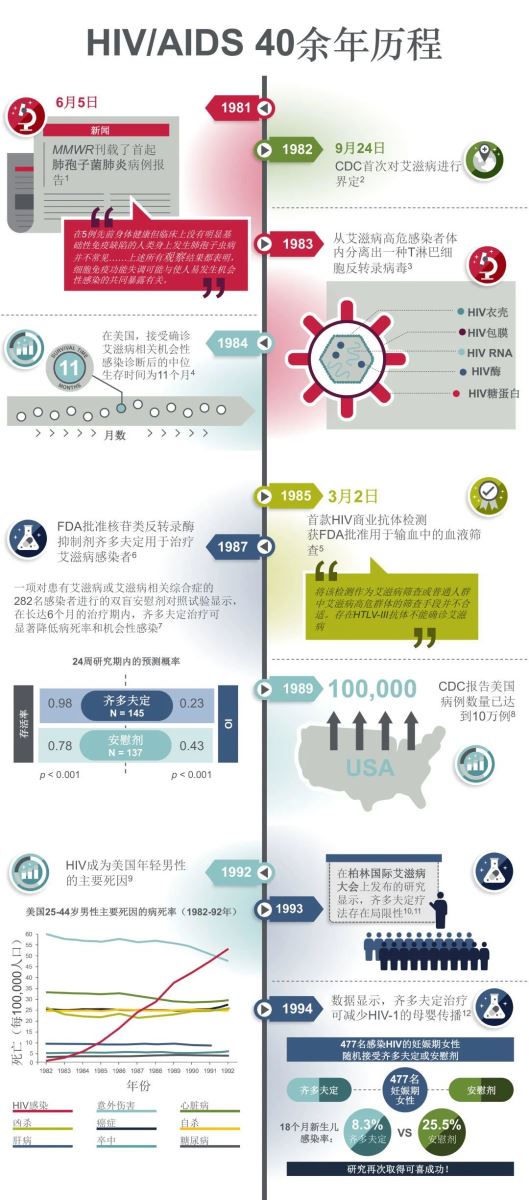
1981年,美国CDC的《发病率与死亡率周报》上报道了5例卡氏肺囊虫病感染病例,成为了有记录的首批HIV感染者,1983年,首次分离出了HIV毒株。
里程碑2. ART时代开启
1987年,核苷类反转录酶抑制剂(NRTI)齐多夫定获批用于治疗HIV感染者,但单一药物难以强效抑制HIV复制。
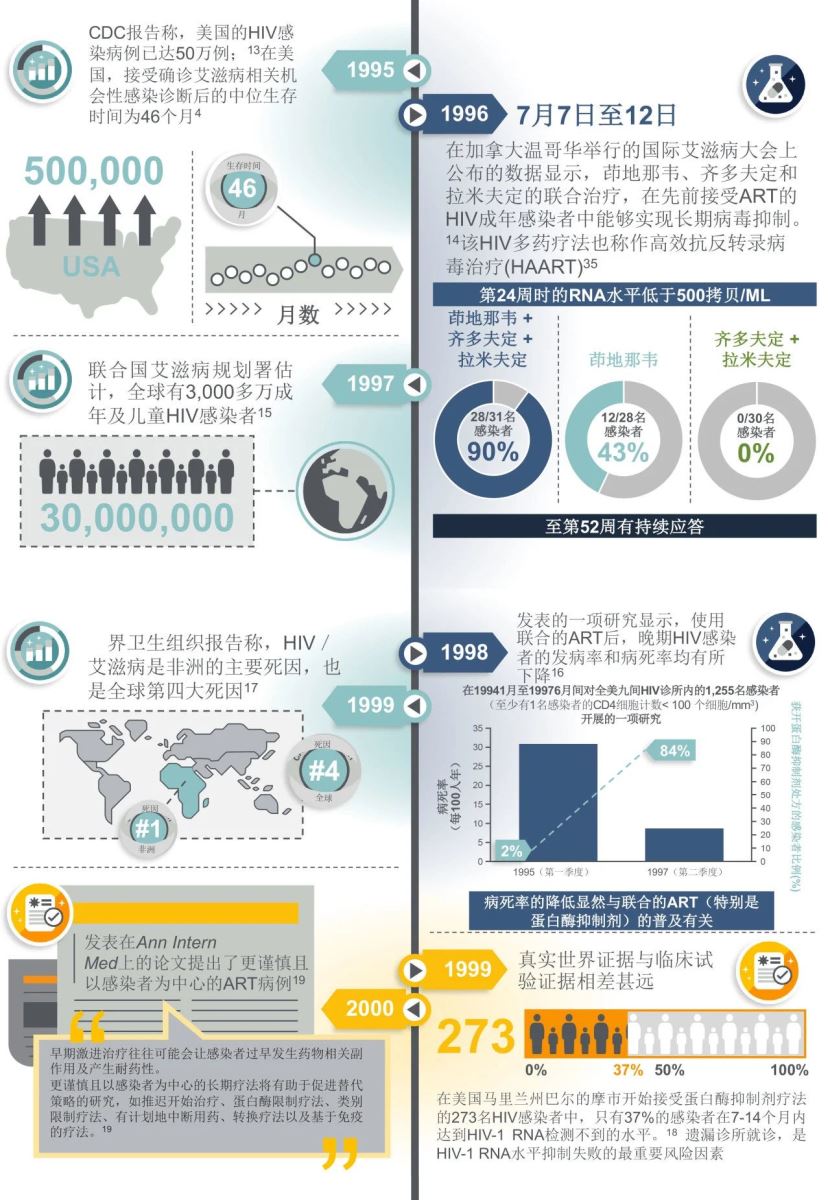
在1996年的第十一届国际艾滋病大会上,报告了茚地那韦、齐多夫定和拉米夫定联合治疗可使HIV感染者的病毒载量长期维持在无法检测的水平,这种多药联合疗法被命名为高活性抗反转录病毒疗法(HAART)。随后,HAART成为了HIV治疗的新标准。
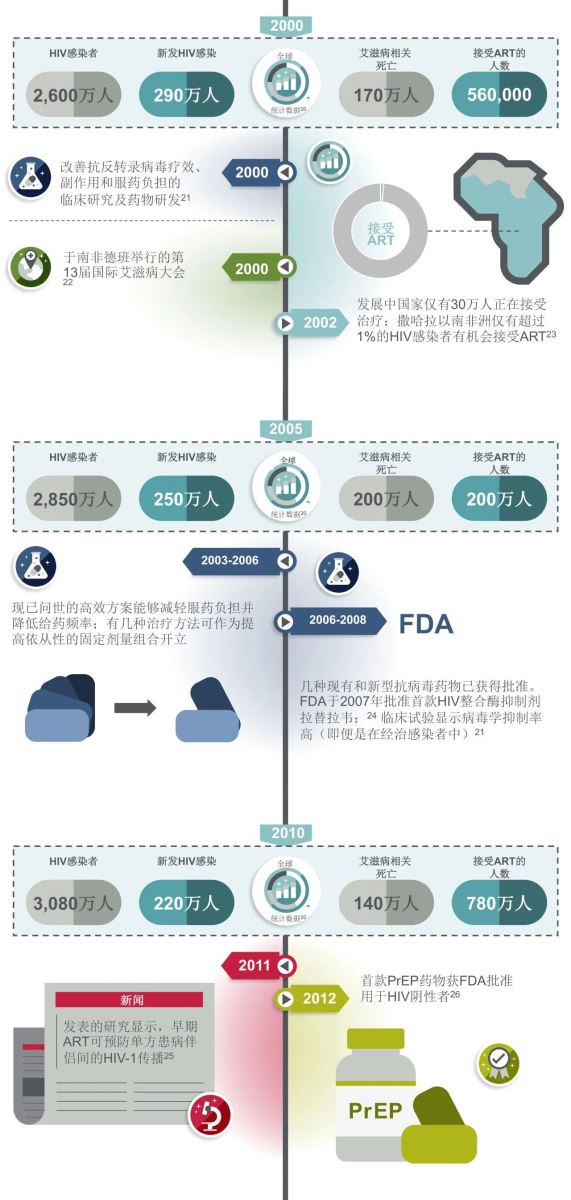
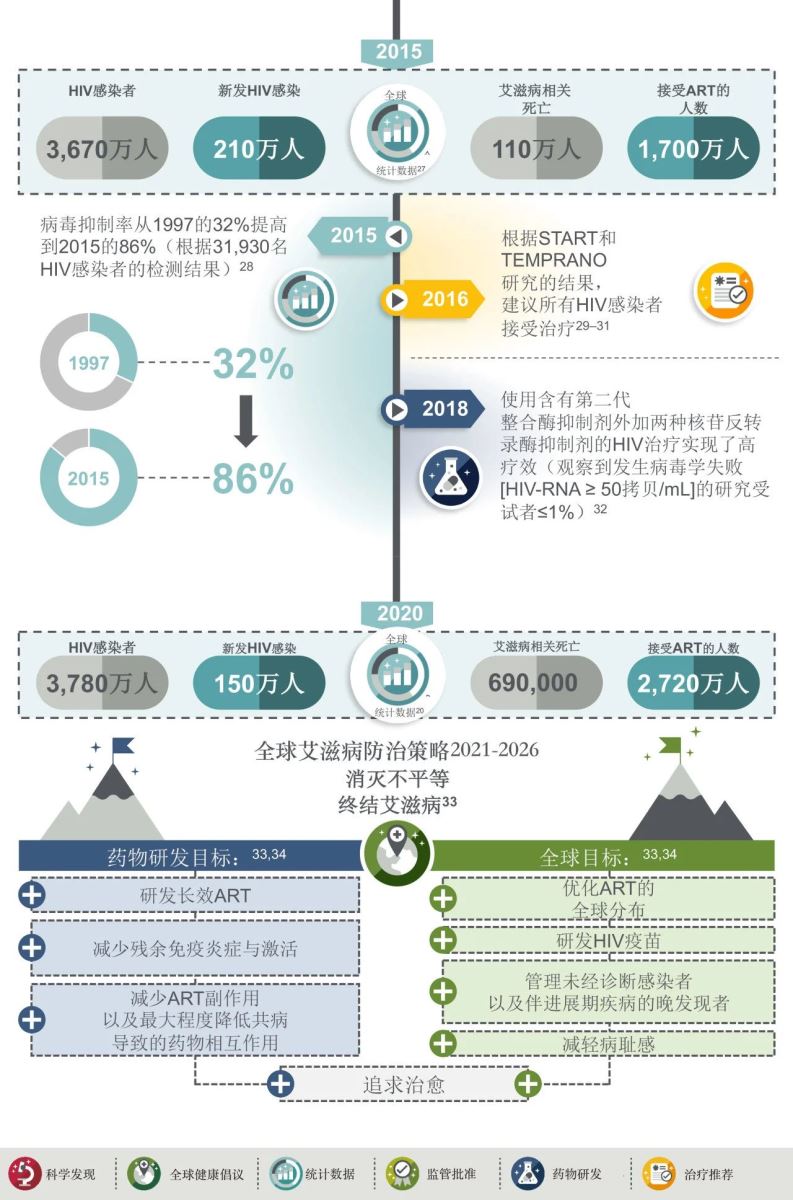
此后的20余年中,ART药物类型得到扩充,蛋白酶抑制剂和整合酶抑制剂带来了更高效、更安全的ART方案,在不同的HIV感染人群中得到验证。2016年,复方单片制剂的出现显著改善了HIV感染者的治疗体验和依从性,具有高耐药屏障的第二代整合酶抑制剂更为感染者提供了可用于快速启动,不受基线因素影响的ART方案。
里程碑3. 确立可行的未来愿景
基于2030年消除HIV流行的目标,一方面,研发新的长效治疗药物,进一步改善HIV感染者的治疗体验,减少残留的免疫炎症和免疫激活,以降低慢性炎症及相关疾病的风险,减少ART药物副作用并使合并症用药带来的药物相互作用最小化;另一方面,弥合ART在全球可及性方面的差距,开发HIV疫苗,加强对未确诊感染者和晚发现者/晚期感染者的管理,减少污名化。在此过程中,寻找治愈方法以消灭HIV,终结艾滋病。
▌参考文献:
[1] UNAIDS. The path that ends AIDS: UNAIDS Global AIDS Update 2023. Accessed: July 2023. Available at: https://thepath.unaids.org/wp-content/themes/unaids2023/assets/files/2023_report.pdf.
[2] Gottlieb M S, Schanker H M, Fan P T, et al. Pneumocystis pneumonia—Los Angeles[J]. Mmwr, 1981, 30(21): 250-2.
来源:《感染医线》
声 明
凡署名原创的文章版权属《感染医线》所有,欢迎分享、转载(开白可后台留言)。本文仅供医疗卫生专业人士了解最新医药资讯参考使用,不代表本平台观点。该等信息不能以任何方式取代专业的医疗指导,也不应被视为诊疗建议,如果该信息被用于资讯以外的目的,本站及作者不承担相关责任。

责任编辑:彭伟彬
相关搜索: HIV